Semana XI
FIS01184
|
|
| |
|
|
|
|
|
|
| |
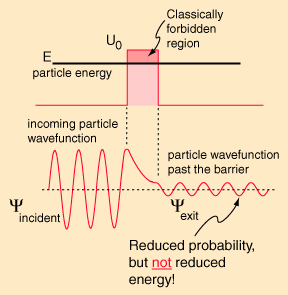
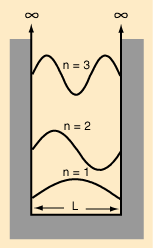
Equação de Schroedinger, Funções de Onda, Densidades de Probabilidade.
|
|
WeeK_LinK:
Função de Onda Equação de Onda
Comprimento de onda de de Broglie
breve introdução à Mecânica Quântica , história da Mecânica Quântica
Nano-computadores (computadores quânticos), Mecânica Quântica
Dualidade
onda-partícula (applets ![]()
![]() )
)
Em
alemão (applets).
teletransporte e experiências físicas relacionadas, A Fun Talk on Teleportation.
Personage_LinK: Erwin Schroedinger